
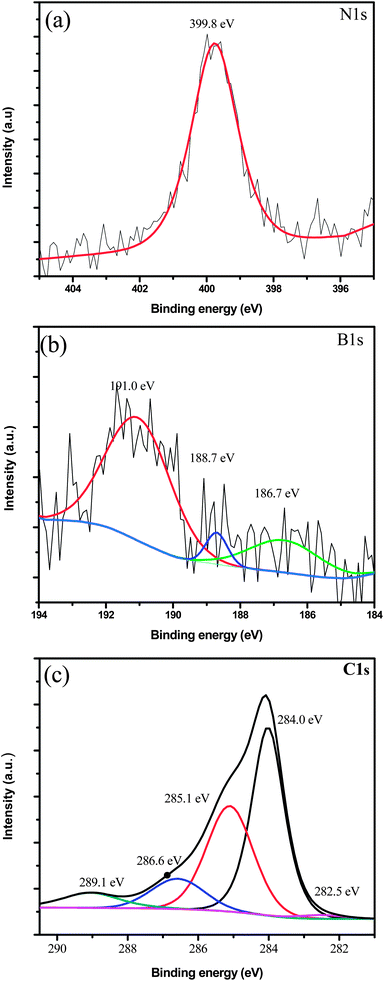
In this work, X-ray Photoelectron Spectroscopy (XPS) was used to study in detail the nitrogen species present in PFPA-functionalized surfaces (PFPA-silane SAMs on silicon wafers as well as PFPA-thiol SAMs on gold slides). 33, 39 The PFPA chemistry has been used to globally modify surfaces or to modify discrete areas by arraying techniques such as photomasking or printing devices. 23, 32– 38 Moreover, the surface density of immobilized molecules can be controlled through the PFPA concentration. The functionalization process is simple and reproducible, and has been proven to be efficient for the immobilization of graphene, proteins and other small biomolecules, carbohydrates and synthetic polymers onto surfaces. 23, 26, 27 This bimolecular reaction, which is much less efficient in alkyl azides or phenyl azides, 28– 31 makes PFPAs highly efficient in surface functionalization. 23– 25 Upon light or thermal activation, the azido group in the PFPA structure is converted to a highly reactive nitrene species that readily inserts into CH and NH bonds or undergoes cycloaddition reaction with C=C bonds. Recently, perfluorophenyl azides (PFPAs) have been developed to introduce functional groups onto solid surfaces.
9– 11 The most common uses of azides are the click chemistry 12– 20 and photochemistry 21, 22. 3, 4, 9– 11 Azide-terminated SAMs provide an excellent route for thin films fabrication and allow tailoring surface properties through covalent immobilization. 1– 8 Self-assembled monolayers (SAMs) have become an excellent technique for the modification and functionalization of a variety of surfaces. The surface structure and composition of materials determine their properties and performance, therefore, tailoring the surface properties of a material through surface modification is an important method to control the function of materials. This study provides strong evidence for the formation of covalent bonds during the PFPA photocoupling process. Finally, XPS analyses were conducted on patterned graphene to investigate the covalent bond formation between the PFPA and graphene. The azide decomposition as the function of x-ray exposure was also determined. The observed 2:1 ratio of the areas from the 402.1 eV to 405.6 eV peaks suggests the assignment of the peak at 402.1 eV to the two outer nitrogen atoms in the azido group and assignment of the peak at 405.6 eV to the central nitrogen atom in the azido group. The three nitrogen peaks detected in the XPS N1s spectra were assigned to amine/amide (400.5 eV) and azide (402.1 and 405.6 eV) species. Here, the challenge of characterizing the PFPA modified surfaces was addressed by detailed XPS experimental analyses. Upon light irradiation, the azido group in PFPA is converted to a highly reactive singlet nitrene species that readily undergoes CH insertion and C=C addition reactions. It is a simple route for the efficient immobilization of graphene, proteins, carbohydrates and synthetic polymers onto a variety of surfaces. PFPA chemistry is a novel immobilization method for tailoring the surface properties of materials. X-ray Photoelectron Spectroscopy (XPS) was used to characterize the nitrogen species in perfluorophenylazide (PFPA) self-assembled monolayers.
